Don’t you love the alliteration of learning all about atoms? If you are teaching middle school science, then your students need to know all about atoms. After all, atoms are the building blocks of matter. There is a lot to know about atoms, but let’s start with what are the three parts of an atom?
What is Matter?
Everything around us is matter. All liquids, solids, and gases are matter. Matter is made up of tiny particles called atoms. The dog, the grass, and even the air in this picture are all matter made up of atoms.
We have to use models to learn about atoms because they are too small to see, even with a microscope.
The History of Atoms
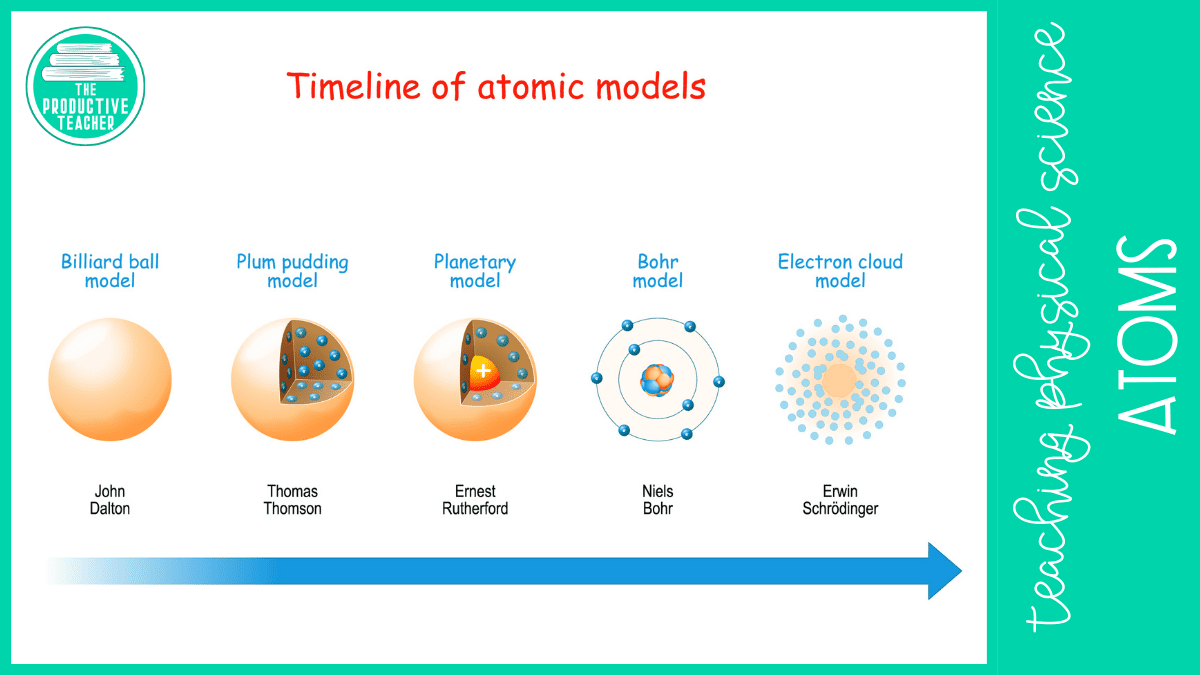
People haven’t always known about atoms. In Ancient Greece, a philosopher named Democritus believed everything on Earth was made up of tiny particles he called atoms. Unfortunately, a more popular Ancient Greek philosopher, Aristotle, disagreed with Democritus, and they were forgotten.
In the early 1800s, an English chemist named John Dalton rediscovered atoms through his experiments with gases. Then, in 1897, JJ Thomson, a physicist from England, discovered negative charges within atoms and developed the plum pudding model. Later, in 1911, Ernest Rutherford, a physicist from New Zealand, found that the negative charges within an atom orbited around a positive center, and in 1913, Neils Bohr updated Rutherford’s model to show that electrons orbited around the nucleus at specific energy levels he called electron shells.
Our current model was updated by Erwin Schrödinger in 1926. He discovered that electrons don’t travel in a set path, but we can model where they are most likely to be found with clouds of probability called orbitals.
What Are the Three Parts of an Atom?
Atoms are too small to see, so we have to use models to think about them.
Our current model has a central nucleus made up of protons and neutrons surrounded by electrons.
Protons and neutrons are the same size, but protons have a positive charge, and neutrons do not have a charge.
Electrons are much smaller than protons and neutrons. Electrons have a negative charge.
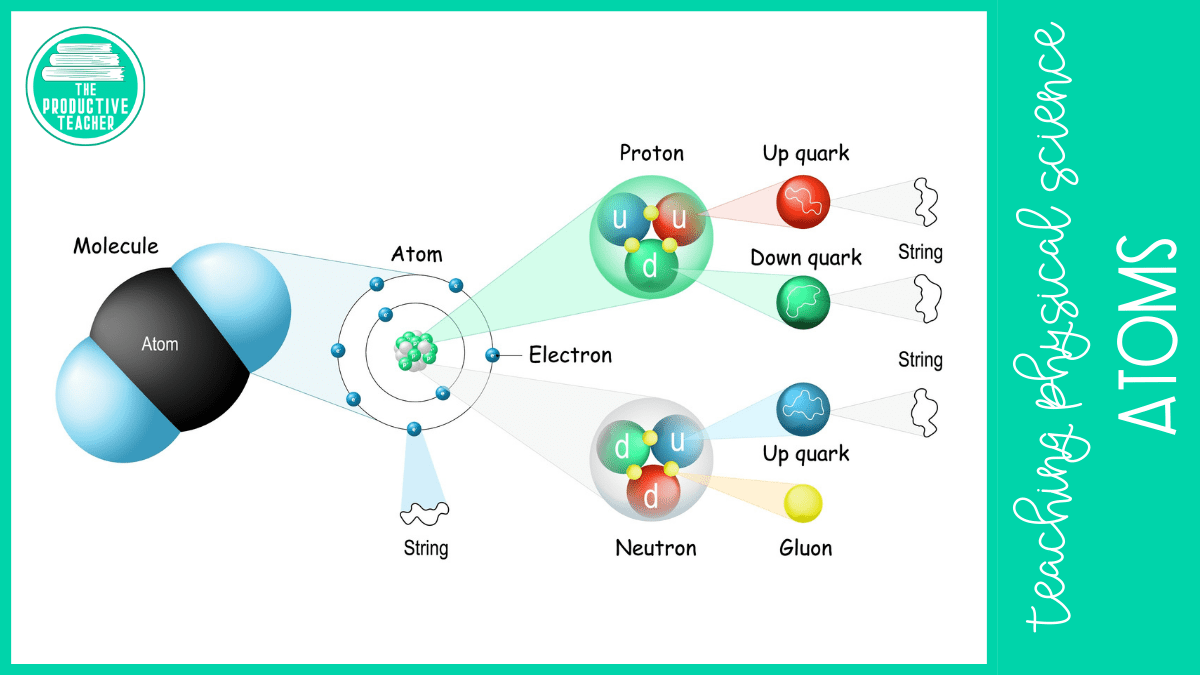
The Smaller Parts of an Atom
Protons and neutrons are made up of even smaller particles called quarks. Scientists believe quarks and electrons are fundamental particles. That means they aren’t made up of anything smaller.
We spend a lot of time learning about the parts of an atom, but 99.99999% of it is empty space. Even protons and neutrons, the largest subatomic particles, are 100,000 times smaller than the atom.
The Electron Cloud
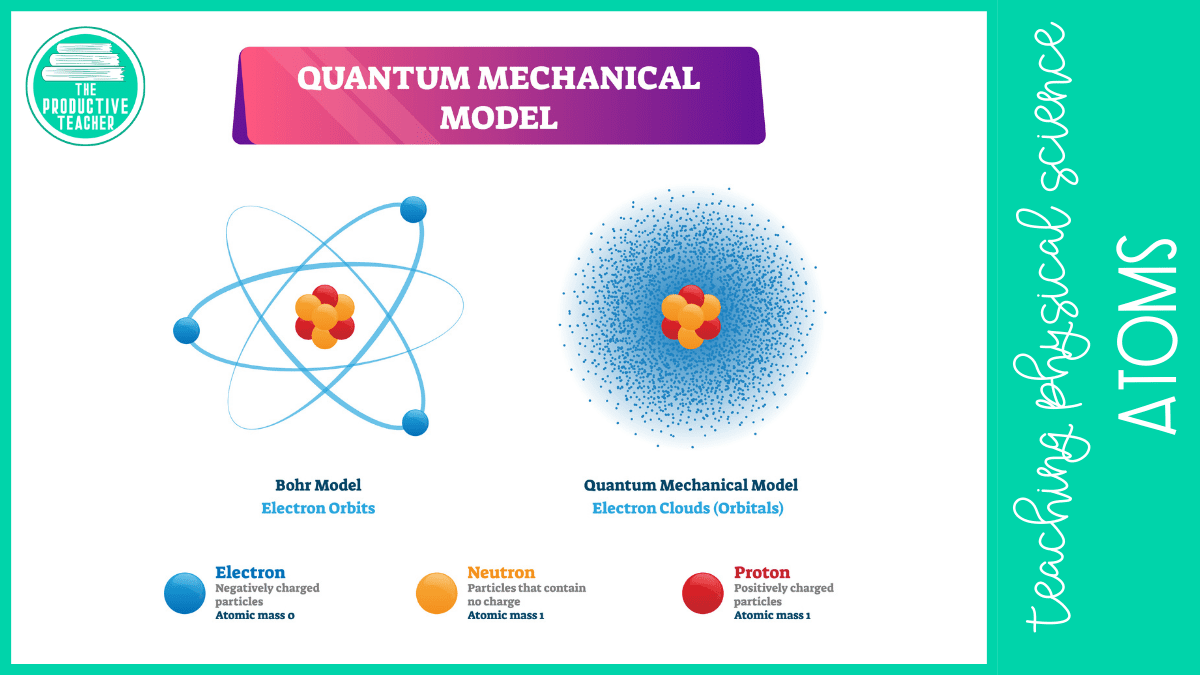
Electrons are 1.800 times smaller than protons and neutrons. These tiny particles move so fast that they do not orbit the nucleus like a planet orbits a star. Instead, they form a negatively charged cloud in the empty space around the nucleus.
Individual negatively charged electrons, like those shown in Bohr’s model, would be attracted to the positively charged protons and fall into the nucleus. The electron cloud can resist this attraction.
Have you ever played with magnets? One side of a magnet will be attracted to another magnet, but the other side of the magnet will repel it. This happens because opposite charges attract each other and like charges repel each other.
The Strong Nuclear Force of Atoms
In an atom, positively charged protons are packed together in the nucleus. They should repel each other and tear the atom apart because they all have the same charge, but they don’t.
Scientists believe a force they call the strong nuclear force holds the protons in a nucleus together. The strong nuclear force is stronger than the force that repels like charges, so the protons in the nucleus stay put.
Updating the Model of Atoms
Our model has changed as people have learned more about it. After Rutherford discovered that electrons orbit around a central nucleus, Niels Bohr refined the model when he discovered that electrons circle the nucleus at specific distances he called orbitals. Later, Erwin Schrödinger found that electrons move so fast that it is impossible to know exactly where an electron is at any given moment, so he updated the atomic model to show a cloud where an electron is likely to be found.
Scientists continue to learn more about the atom. The neutron wasn’t even discovered until 1932! Then, in 1964, scientists discovered the quarks that make up protons and neutrons. As we learn more, our atomic models will change.
Atoms and Elements
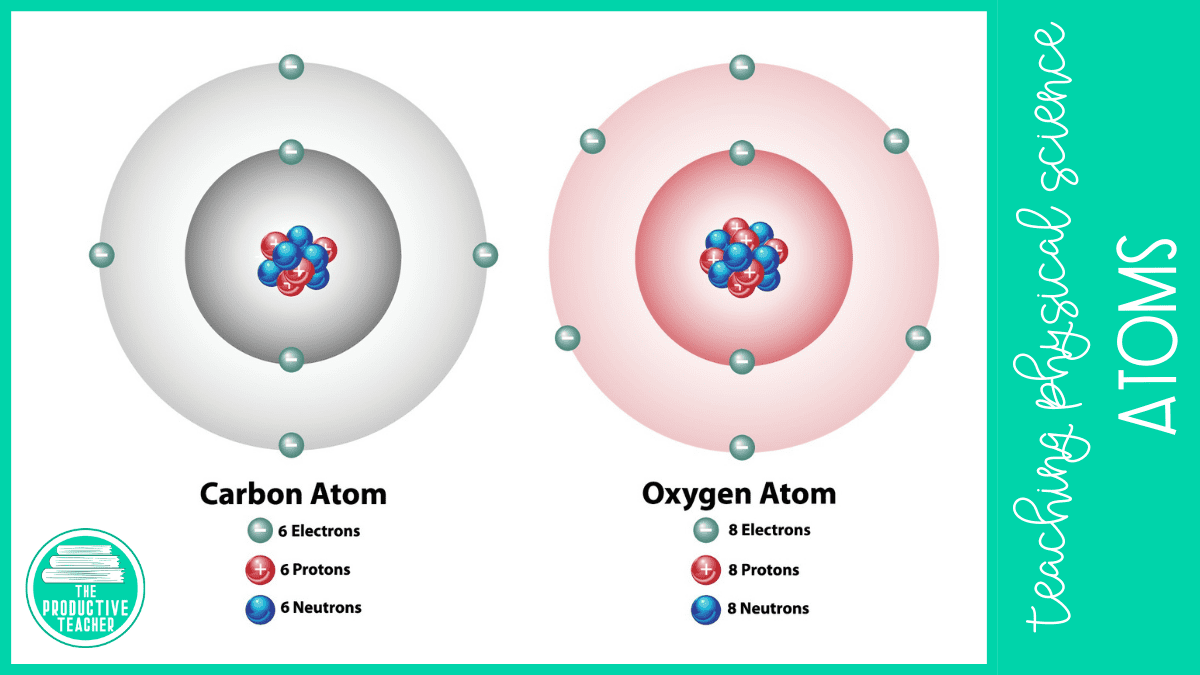
Everything around us is made up of atoms, but they aren’t all the same. Atoms have different numbers of protons, neutrons, and electrons.
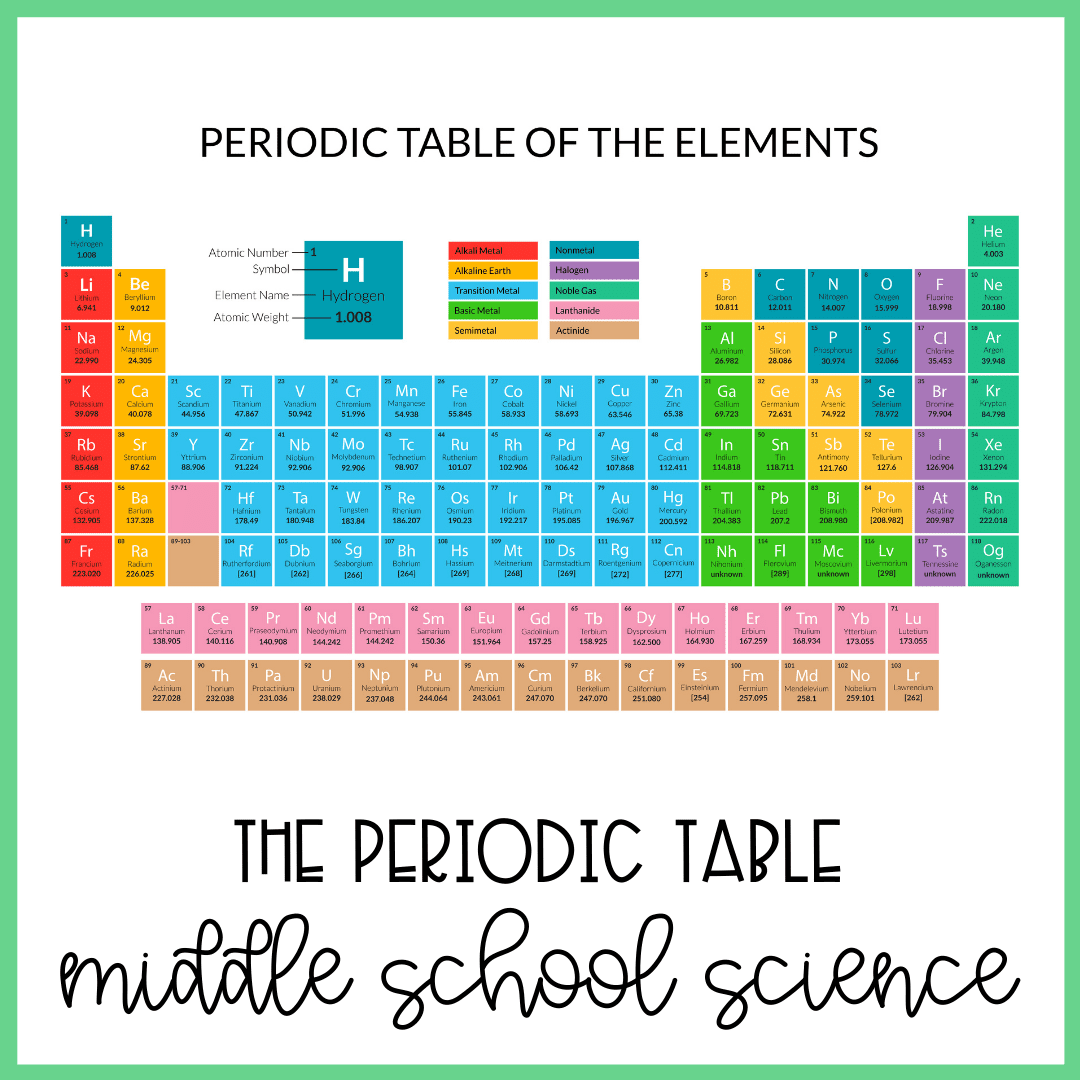
An element is a substance made up of one type of atom. For example, hydrogen is an element. Every atom of hydrogen has one proton and one electron. Carbon is another element. Every atom of carbon has six protons, six neutrons, and six electrons. Scientists organize the elements in the periodic table of the elements.
Digital Science Unit
If you want to learn about atoms, you have to do more than read about them one time. That’s why my digital science unit has review activities, flashcards, and comprehension questions. If you want to learn something, you have to build your neural network, and that takes practice. You can see every page of my digital science unit in the video below.
Want to learn more? Check out my atoms digital science unit at Teachers Pay Teachers. Click the picture below!
ARE YOU TEACHING ANOTHER SCIENCE TOPIC?
I am working on creating more science units so that every science teacher can get exactly what he or she needs for her students. You can also read about how I use brain science to teach other science topics on my blog. Click the pictures below to learn more.











Leave a Reply