If we want to understand the modern periodic table, we have to understand matter first.
Matter and the Modern Periodic Table
Everything around us is matter. Atoms make up all matter. An atom has a central nucleus containing protons and neutrons. Negatively charged electrons spin around the nucleus.
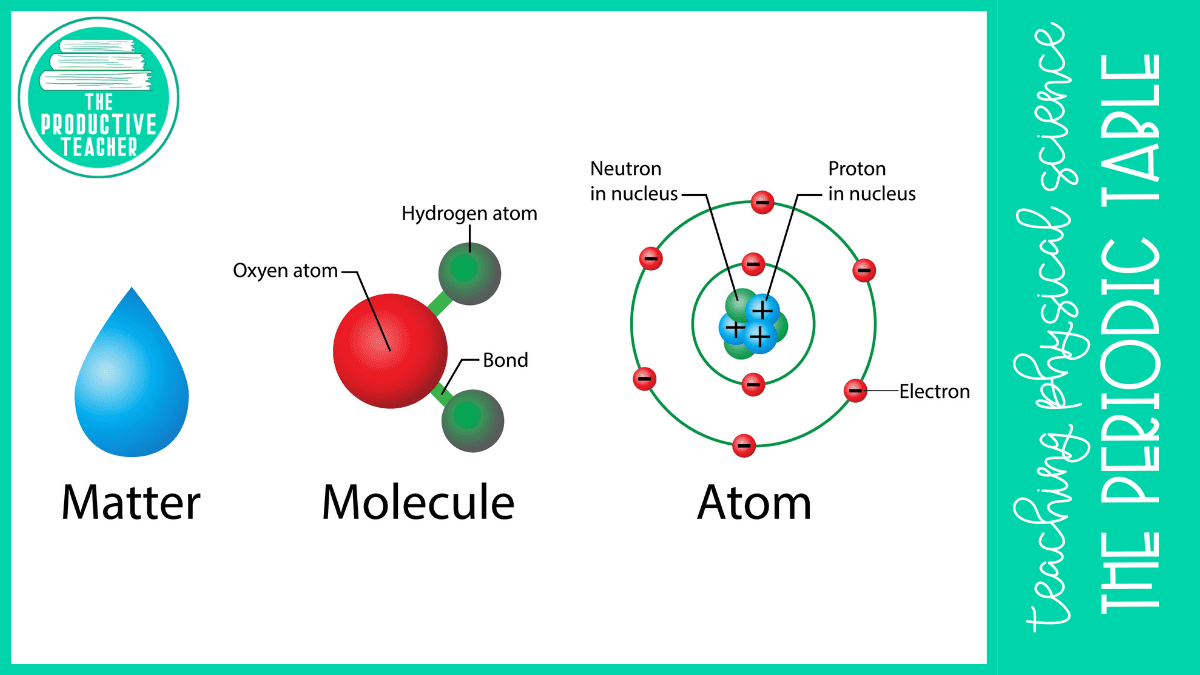
Elements are substances made up of one type of atom. Every atom of an element has the same number of protons. For example, every carbon atom has six protons. Every hydrogen atom has one proton.
Not every substance is an element. For example, water is not an element. It is a compound because it is made up of both hydrogen and oxygen atoms. Other compounds include table salt and sugar.
Scientists use the modern periodic table to organize all of the known elements.
The History of the Modern Periodic Table
Chemists have been studying the substances around them for thousands of years, but didn’t discover first element until 1669 when Hennig Brand isolated phosphorus from urine. As an alchemist, Brand searched for a way to make gold using a variety of creative resources.
In 1803, the English chemist John Dalton created the first periodic table to organize the known elements. His first table had just five elements. As he learned about more elements, he added them to his periodic table.
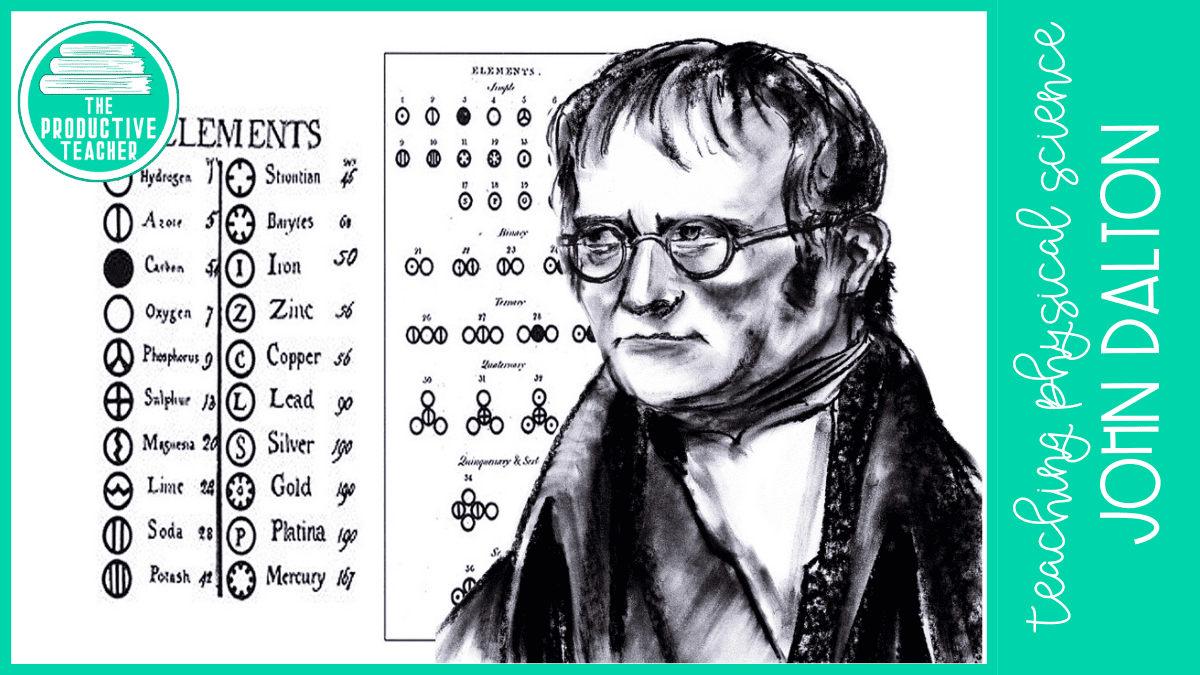
By the 1820s, many new elements had been discovered. Jöns Jakob Berzelius, a chemist from Sweden, made an updated periodic table. He used abbreviations for the elements based on their Latin names. We still use Berzelius’s abbreviations today. For example, gold’s symbol on the periodic table is Au because gold is called aurum in Latin.
Dmitri Ivanovich Mendeleev
By 1860, there were over 60 known elements. A Russian chemistry professor, Dmitri Ivanovich Mendeleev, was writing a textbook and wanted a way to organize all of the known elements for his students. He wrote the names of the known elements on cards and put the cards up on his wall. He spent almost a year organizing and reorganizing the cards.
Ultimately, he decided to arrange the elements according to atomic weight. Hydrogen, the lightest element, was first. When he finished, his table showed even more patterns. For example, the metals and nonmetals were on opposite sides of the table. He put elements with similar properties in columns he called groups. Mendeleev needed to leave some empty spaces to make his table work, but he predicted that other elements would fill in the holes. By 1875, chemists had discovered three more elements. Each of them fit within Mendeleev’s table.

Today, we still use Mendeleev’s periodic table. There are currently 118 elements on the periodic table, but scientists believe there are still more elements to discover.
Atomic Number and the Modern Periodic Table

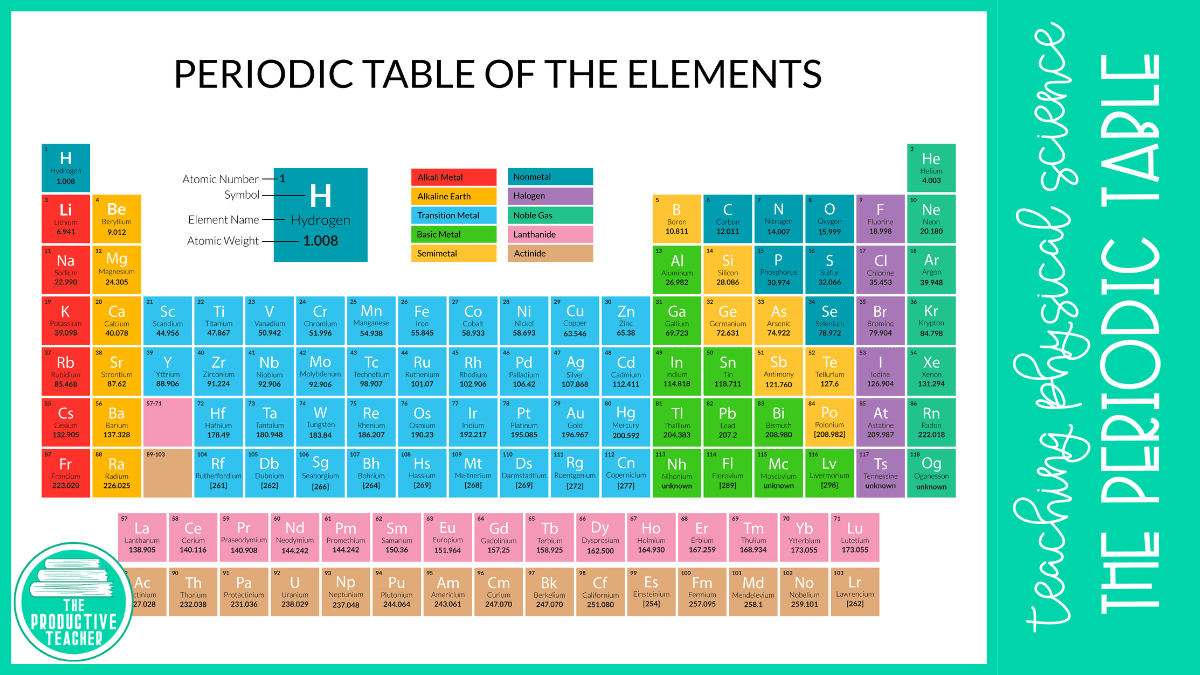
The periodic table we use today is organized by atomic number. An element’s atomic number is the number of protons in one atom. For example, hydrogen has an atomic number of 1 because each atom of hydrogen has one proton.
There are 7 rows called periods in the periodic table. Each period begins a new energy level for the electrons that orbit around the nucleus in atoms.
The periodic table also has 18 columns called groups or families. All of the elements in a group have similar properties because they have the same number of electrons in the outermost energy levels or shells. For example, both oxygen and sulfur have six electrons in their outermost energy levels or shells.
Element Cards
Each element on the periodic table is represented by a card like the one shown on this page. The card shares information about the element.
The top number shows the number of protons in the atoms of the element. For example, carbon has six protons. In a neutral atom, the number of protons equals the number of electrons. So, carbon also has six electrons.
The large C is the chemical symbol for carbon. Scientists use this symbol in chemical equations. You can see carbon’s name under the chemical symbol.
The bottom number is the average atomic mass of the element. While atomic mass is always a whole number because atomic mass is equal to the number of protons and neutrons in an atom, not all atoms have the same number of neutrons. Some carbon atoms have 6 neutrons, while others have 5 or 7. We call atoms with different numbers of neutrons isotopes. The average atomic mass is the average of all of the isotopes based on their abundance in nature.
Atoms and the Modern Periodic Table
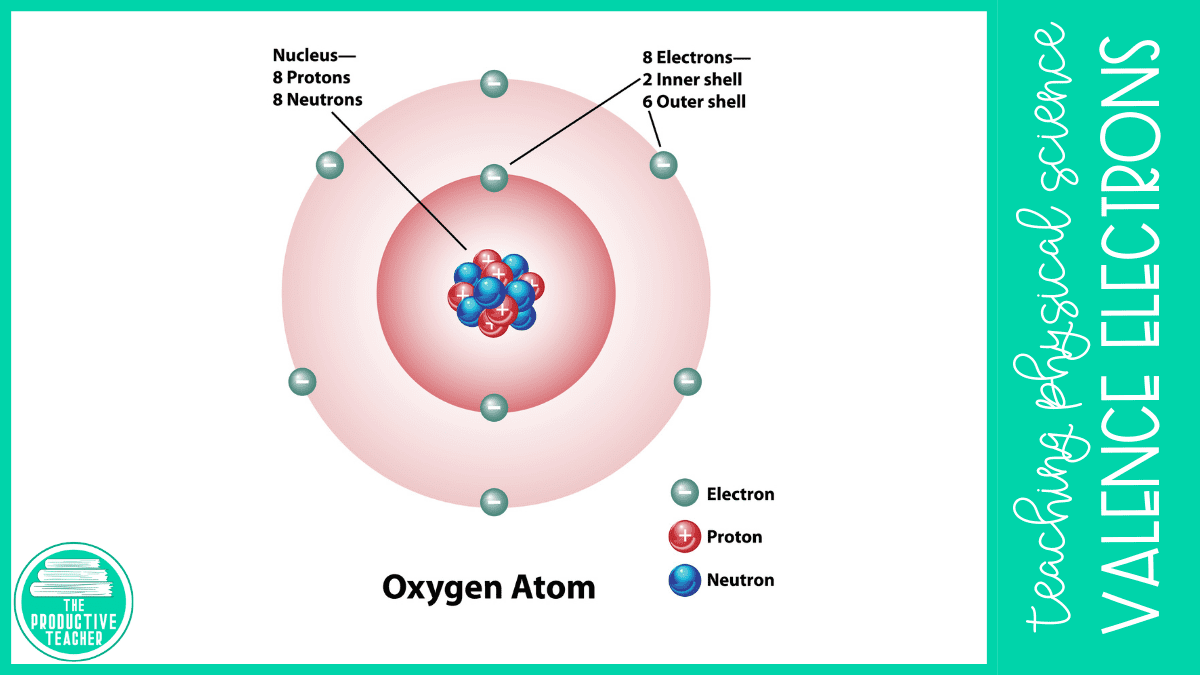
Atoms have a central positively charged nucleus made up of protons and neutrons. The protons have a positive charge of +1, and the neutrons do not have a charge. Negatively charged electrons surround the atom’s nucleus. Electrons have a negative charge of -1.
Electrons orbit around the nucleus in distinct energy levels called electron shells. We label electron shells with numbers starting at 1. The higher the number, the farther the electrons are from the nucleus and the more energy they have.
We call the outermost shell of electrons the valence shell, andtThe electrons within the shell valence electrons. To be stable, atoms must have a full valence shell. They will give up or take electrons from other atoms to fill their valence shells. Most elements need 8 electrons to fill their atoms’ valence shells, but elements in the first two groups only need two electrons.
Groups in the Modern Periodic Table
The elements in the groups of the periodic table have similar properties. They interact with other elements in similar ways because they have the same number of electrons in their outer shells.
We call the first group alkali metals. These elements are extremely reactive because they only have one electron in their outer shells, so they are eager to give it up.
We call the second group alkaline earth metals. These elements are also very reactive because they only have two electrons in their outer shells.
Groups 3 through 12 are transition metals. These elements fill their two outer shells slightly out of order. As a result, they share electrons differently from the other groups.
Group 13 is also known as Group 3A because these elements have three electrons in their valence shells.
The elements in Group 14, or Group 4A, have four electrons in their valence shells. Carbon, an element in Group 14, can share all four of its valence electrons to make a wide variety of compounds.
Group 15, 16, and 17 follow the same pattern with five, six, and seven electrons in the valence shells. Atoms from these groups are more likely to add electrons than give them up.
Group 18, or Group 8A, are the noble gases. The atoms in these elements all have full valence shells, so they do not lose or gain electrons.
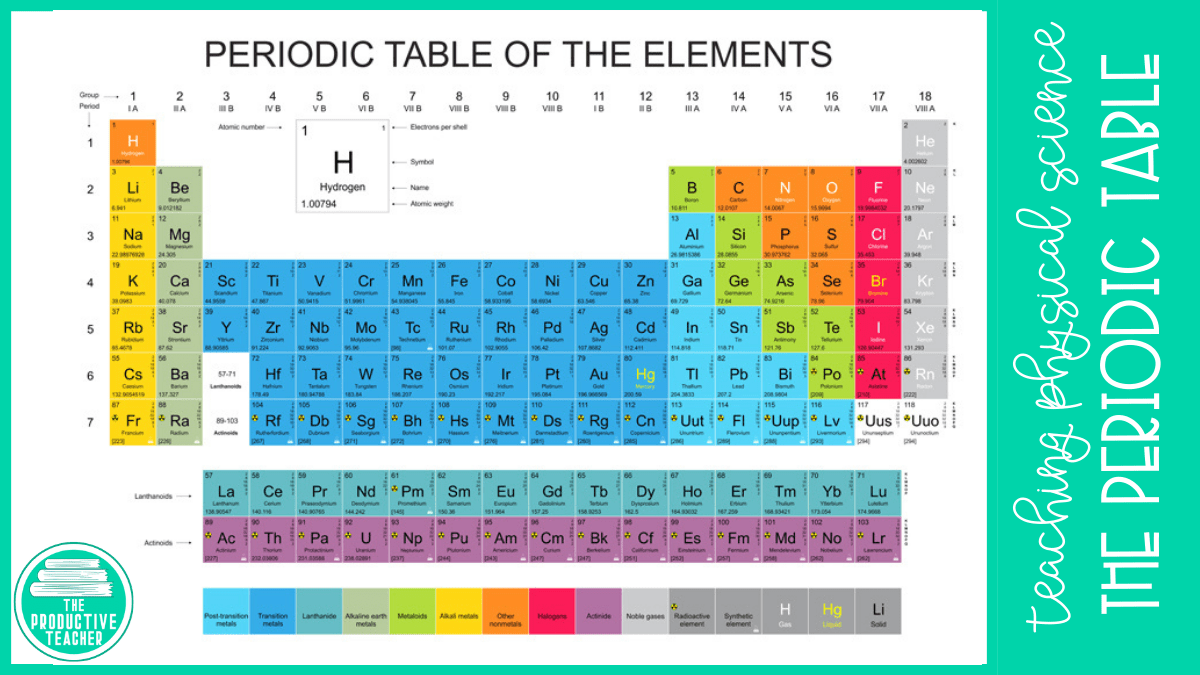
Lanthanides and Actinides
The lanthanides and actinides make up the two rows of elements underneath the rest of the periodic table. These elements behave differently from the other transition metals because of the way their electrons fill the electron shells.
We also call Lanthanides rare-earth or inner transition metals. We find all of these elements in nature. Promethium is the only radioactive lanthanide.
On the other hand, all actinides are radioactive, including uranium and plutonium, which are used in nuclear bombs. We find some of the actinides in nature, but others have only been observed in a laboratory.
Understanding the modern periodic table is key for any chemistry student because so much information is hidden in the table. For example, chemists can predict how elements will react based on their positions on the table. Do you want to learn more about the modern periodic table? Then you need my periodic table digital science unit. It has all of the information in this blog post and pictures, video links, activities, flashcards, and comprehension questions. You can see all of the pages in the unit in the video below.
As you can see, this digital science unit will help you master the basic information on the modern periodic table. You can click the picture below to check out the resource at Teachers Pay Teachers.
ARE YOU TEACHING ANOTHER SCIENCE TOPIC?
I am working on creating more science units so that every science teacher can get exactly what he or she needs for her students. You can also read about how I use brain science to teach other science topics on my blog. Click the pictures below to learn more.











Leave a Reply